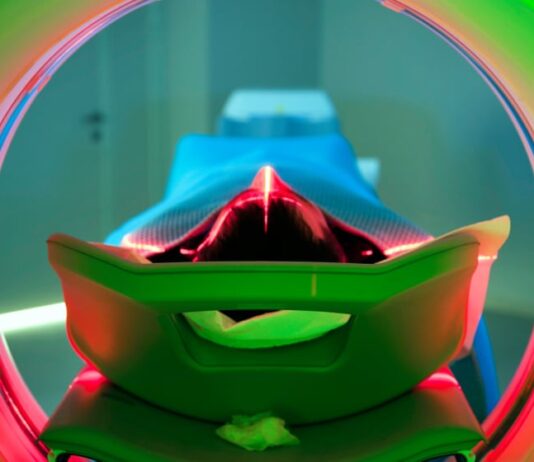
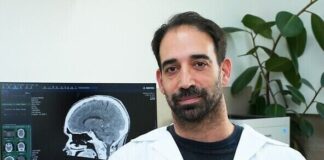
In breakthrough, Israeli scientists use deep-brain stimulation to counter schizophrenia
Novel study by researchers at the Rambam Health Care Campus could help 21 million people worldwide suffering from the severe lifelong mental disorder
Source link...
Sepsis ‘breakthrough’: the world’s first targeted treatment?
Sepsis is one of the leading causes of death and disability worldwide. It can affect anyone, and is notoriously difficult to diagnose in the...
Two Women Engineers Celebrated by Sony and Nature for Breakthroughs in
Two Pioneering Women from University of Michigan Engineering Receive Prestigious Sony Women in Technology Award
In a groundbreaking achievement, two of the three recipients of...
New Zealand Breakthrough Brings New Hope for Lymphoedema Treatment
A groundbreaking study published in the prestigious journal Cell Reports has illuminated the critical role of insulin-like growth factor (IGF) signaling in the development...
Pill Breakthrough in Early Parkinson’s Treatment
UConn School of Medicine offering innovative clinical trial testing novel pill's impact for reducing inflammation to treat Parkinson's diseaseBill is only in his fifties,...
Lab grown human spinal cord heals after injury in major breakthrough
Scientists at Northwestern University have created the most sophisticated lab grown model yet for studying human spinal cord injury.
In the new research, the team...
IBD’s New Power Rankings for 2026: The 5 Breakthrough Remedies Redefining...
The inflammatory bowel disease (IBD) treatment market is undergoing one of its most rapid evolutions in decades, driven by biologic innovation, targeted immune therapies,...
This Breakthrough Therapy ‘Reprograms‘ Cells to Regrow Cartilage
The squishy insoles in your favorite pair of sneakers help keep your joints safe from daily wear-and-tear, but over time, that cushioning wears down....
Biotech Company Advances Breakthrough Hyperinsulinism Treatment
Rezolute Inc. (RZLT:NASDAQ), a late-stage company focused on treating hypoglycemia caused by all forms of hyperinsulinism (HI), reported financial results and...
Scientific Wonders In The Field Of Health(2024-2026)
Here is an overview of new discoveries in the field of health (2024–2026), highlighting breakthroughs in medicine, biotechnology, neuroscience, and public health.🧬 1. Gene...